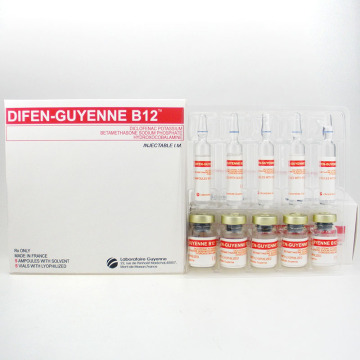
Vitamin B12 Difen-Guyenne B12 Diclofenac Potassium Betamethasone Sodium
Basic Info
Model No.: vitamin b12
Product Description
Model NO.: vitamin b12
Usage Mode: For external use
State: Solid
Type: Organic Chemicals
Trademark: GUYENNE
Origin: France
Application: Internal Medicine
Suitable for: Elderly, Adult
Shape: Powder
Pharmaceutical Technology: Chemical Synthesis
Specification: GMP/WHO GMP/EURO GMP
HS Code: 3001901000
vitamin b12 Difen-Guyenne B12 Diclofenac Potassiumbetamethasone Sodium Hydroxocobalamine
Product Description
DIFEN-GUYENNE B12 DICLOFENAC POTASSIUMBETAMETHASONE SODIUM
Diclofenac Potassium - Clinical Pharmacology
Pharmacodynamics
Diclofenac Potassium is a non-steroidal anti-inflammatory drug (NSAID) that exhibits anti-inflammatory, analgesic, and antipyretic activities in animal models. The mechanism of action of Diclofenac Potassium, like that of other NSAIDs, is not completely understood but may be related to prostaglandin synthetase inhibition.
Pharmacokinetics
Absorption
Diclofenac is 100% absorbed after oral administration compared to IV administration as measured by urine recovery. However, due to first-pass metabolism, only about 50% of the absorbed dose is systemically available (see Table 1). In some fasting volunteers, measurable plasma levels are observed within 10 minutes of dosing with Diclofenac Potassium. Peak plasma levels are achieved approximately one hour in fasting normal volunteers, with a range of .33 to 2 hours. Food has no significant effect on the extent of diclofenac absorption. However, there is usually a delay in the onset of absorption and a reduction in peak plasma levels of approximately 30%.
Special Populations
Pediatric
The pharmacokinetics of Diclofenac Potassium has not been investigated in pediatric patients.
Race
Pharmacokinetic differences due to race have not been identified.
Hepatic Insufficiency
Hepatic metabolism accounts for almost 100% of Diclofenac Potassium elimination, so patients with hepatic disease may require reduced doses of Diclofenac Potassium compared to patients with normal hepatic function.
Renal Insufficiency
Diclofenac pharmacokinetics has been investigated in subjects with renal insufficiency. No differences in the pharmacokinetics of diclofenac have been detected in studies of patients with renal impairment. In patients with renal impairment (inulin clearance 60 to 90, 30 to 60, and < 30 mL/min; N = 6 in each group), AUC values and elimination rate were comparable to those in healthy subjects.
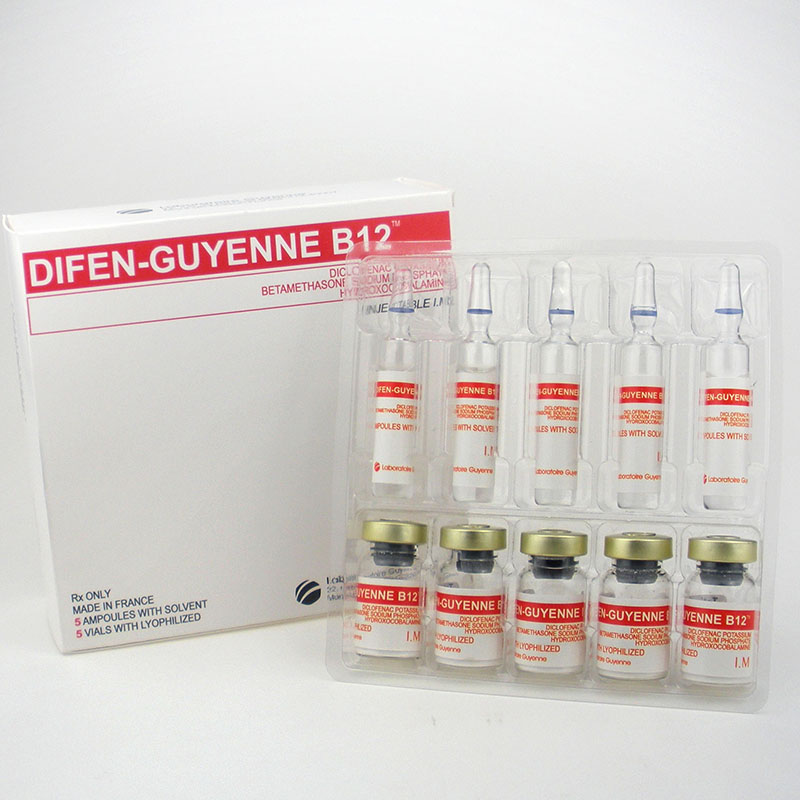
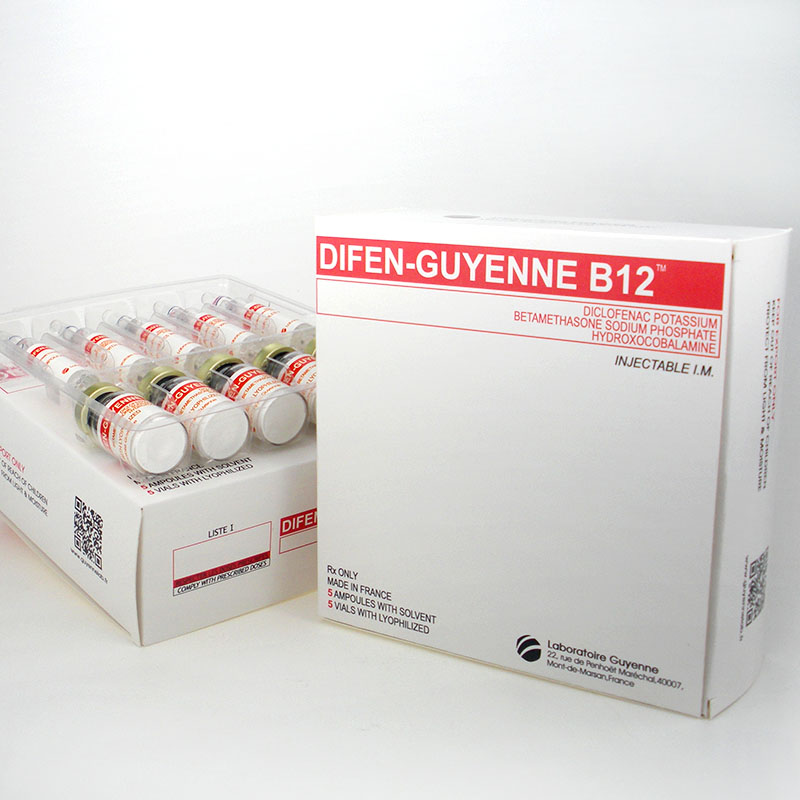
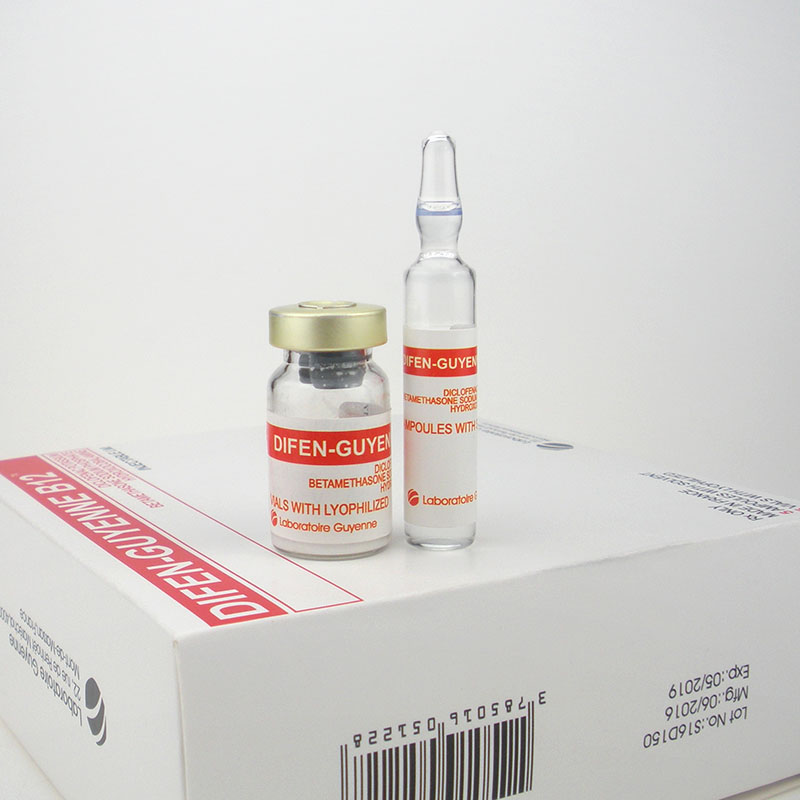
Contact us if you need more details on Difen-Guyenne B12. We are ready to answer your questions on packaging, logistics, certification or any other aspects about Dicolfenac Potassium、Betamethasone Sodium Phosphate. If these products fail to match your need, please contact us and we would like to provide relevant information.
Product Categories : Antipyretic & Analgesic Drug
Premium Related Products
Other Products
Hot Products
High Quality 200mg Amiodarone Hydrochloride TabletsLarge Stock Whiten Skin Monobenzone CreamGeneral Medicine Omeprazole 20mg Injection for Gastrohelcosis and Stomach AcidGeneral Medicine Ceftriaxone Sodium InjectionReady Stock Skin Whitening Anti-Aging Vitamin C InjectionOEM Tablet 500mg ParacetamolHigh Quality 500mg Hard Capsules Amoxicillin (amoxycillin)Best and Low Price Ceftriaxone Sodium Injection CeftriaxoneBest Quality Omega 3 Deep Sea Softgel Capsule Fish OilFDA Approved Curative Antimalarial ArtemisininMedicine Treating Brain Injury Citicoline Sodium InjectionProtease Alpha Chymolase for Inflammatory EdemaGeneral Medicine Medroxyprogesterone Acetate InjectionAmikacin Injection General Medicine DrugsSkin Whitening Gsh Personal Care Glutathione InjectionBody Slimming Fitness Lose Weight Weight Loss L-Carnitine Injection2.0g/5ml